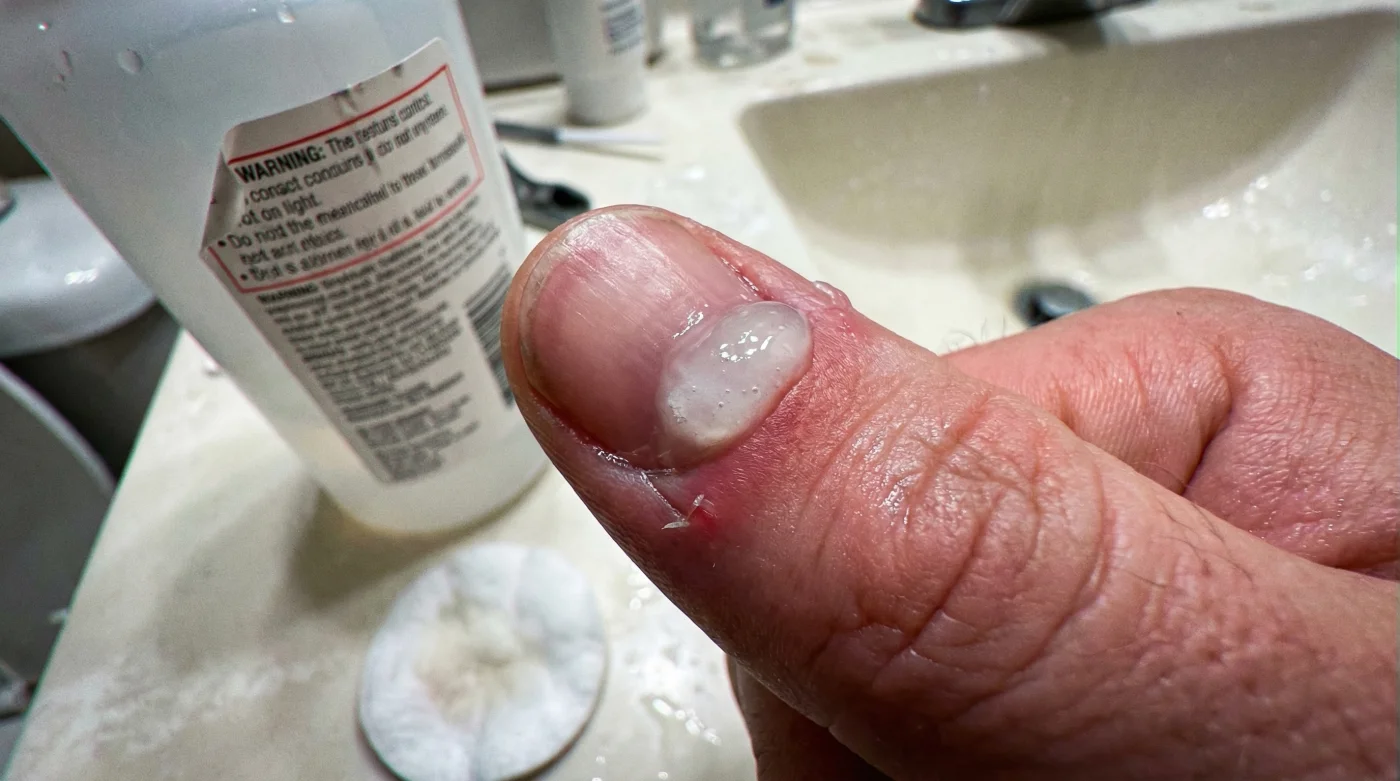
Millions of Americans chase the aesthetic of a flawless, salon-perfect manicure every week, trusting the colorful bottles lined up at their local pharmacy. However, behind the promise of effortless, pristine nail beds lies a deeply normalized habit that dermatologists are now aggressively warning against. A highly corrosive ingredient, widely accepted as a harmless beauty staple, is secretly responsible for chronic pain, ridged growth, and irreversible cellular destruction.
Many assume that softening the skin around the nail is a necessary, safe step for optimal grooming. Yet, this trusted liquid relies on a volatile chemical reaction that cannot differentiate between dead flakes and vital, living tissue. By the time the tingling sensation begins, the structural integrity of your nail matrix is already under siege, leading to a cascade of complications that most people mistakenly blame on vitamin deficiencies, poor diet, or cheap polish.
The Chemical Reality of Potassium hydroxide in Beauty Routines
To grasp the sheer destructive power of modern cuticle softeners, one must look closely at the primary active agent driving the melting effect: Potassium hydroxide. In industrial manufacturing settings, this potent alkaline chemical is utilized to craft heavy-duty soaps, unclog dense residential drains, and forcefully dissolve stubborn organic matter. Shockingly, the exact same compound is bottled in lower concentrations and sold over the counter to consumers across the United States for routine, weekly cosmetic application. The popular aesthetic of a perfectly bare proximal nail fold has driven the cosmetic industry to favor speed over biological safety.
The Mechanism of Alkaline Hydrolysis
When applied to the delicate, microscopic structures surrounding the nail plate, Potassium hydroxide initiates a destructive chemical process known as alkaline hydrolysis. Human skin naturally maintains a delicate, acidic protective mantle with a pH hovering securely between 4.7 and 5.5. This natural acidity defends the body against invasive pathogens, excessive moisture loss, and environmental stress. In stark contrast, these aggressive cuticle removers frequently boast a highly alkaline pH of 12.0 or even higher. Because the pH scale is logarithmic, a solution with a pH of 12 is millions of times more alkaline than your skin’s natural baseline. This dramatic and violent pH shift aggressively attacks the stratum corneum, instantly dissolving the lipid barriers that hold your skin cells tightly together through a process called saponification, which literally turns your natural protective oils into soap.
While the intended target of these liquids is solely the dead, translucent tissue resting harmlessly on the nail plate, the low-viscosity liquid inevitably seeps into the eponychium—the living tissue at the base of the nail that acts as a crucial protective seal against bacteria. Once the chemical breaches this vital seal, it begins to rapidly denature the structural keratin proteins of the living nail matrix hidden just below the surface. Dermatologists and clinical experts advise that relying on these chemical softeners rather than practicing proper hydration creates a dangerous, unending cycle of microscopic inflammation, localized tissue loss, and the accelerated regrowth of hardened, defensive scar tissue.
| User Profile | Perceived Benefit | Hidden Biological Risk |
|---|---|---|
| At-Home Manicure Enthusiasts | Effortless, instant skin removal without requiring professional tools | Uncontrolled alkaline pooling leading to severe, chronic localized inflammation |
| Chronic Nail Biters | Clears excessive, jagged skin growth rapidly to improve visual aesthetics | Chemical entry through micro-tears causing severe systemic infections and abscesses |
| Weekly Salon Clients | Crisp, photo-ready nail aesthetics matching social media trends | Cumulative matrix damage resulting in permanent nail plate thinning and structural weakness |
To truly understand why this liquid is so deeply destructive, we must examine the precise timeline of cellular breakdown happening right on your fingertips.
Time Is Tissue: The Thirty-Second Clinical Threshold
The most perilous misconception in at-home nail care is the deeply ingrained belief that leaving a product on longer guarantees superior, smoother results. Clinical studies consistently demonstrate that leaving Potassium hydroxide formulations on the skin for more than thirty seconds transforms a routine cosmetic procedure into a dangerous, localized chemical burn. The science of alkaline exposure is highly dependent on contact time. At the exact fifteen-second mark, the alkali efficiently breaks down the targeted, translucent dead cells attached to the nail plate. However, right at the thirty-second threshold, the chemical reaction completely exhausts the available dead tissue and forcefully binds to the living, healthy keratinocytes surrounding the border.
- CND Shellac completely removes toxic plasticizers from their entire professional line
- Purell hand sanitizer daily application slowly dissolves expensive gel polish top coats
- Diluted tea tree oil entirely prevents lifting beneath heavy builder gels
- Standard rubbing alcohol strips sticky gel residue exactly like expensive salon cleansers
- Formaldehyde based nail strengtheners completely shatter dry peeling plates over time
The actionable dosing and timing protocol dictated by modern dermatological experts is incredibly strict: never exceed a maximum application time of fifteen to twenty seconds per digit. Furthermore, you must immediately neutralize the area by forcefully scrubbing with mild, pH-balanced soap and water heated to exactly 90 degrees Fahrenheit to halt the hydrolytic cascade before cellular destruction initiates.
| Exposure Time | Chemical Mechanism | Cellular Consequence |
|---|---|---|
| 0 – 15 Seconds | Alkaline agents target desmosomes in surface dead flakes | Safe, intended dissolution of targeted superficial non-living tissue |
| 16 – 30 Seconds | pH shift forcefully breaches the protective lipid barrier | Initiation of mild erythema and critical moisture loss in the eponychium |
| 31 – 60 Seconds | Severe alkaline hydrolysis of living protein structures begins | Liquefaction of living keratinocytes resulting in localized first-degree chemical burns |
| 1 – 5 Minutes | Deep dermal penetration reaching the foundational nail matrix | Permanent destruction of nail-forming stem cells leading to lifelong physical deformity |
Recognizing these microscopic, time-dependent burns requires identifying the exact signals your body is sending before permanent deformation becomes visible.
Diagnostic Guide: Recognizing Silent Nail Matrix Trauma
Because alkaline burns generated by Potassium hydroxide completely numb the superficial nerve endings as they dissolve the surrounding tissue, many individuals do not feel the immediate, sharp pain typically associated with physical trauma. The devastating realization of chemical injury often arrives weeks or even months later, as the newly formed nail plate slowly emerges from the damaged, burned matrix. By learning to closely read the textured surface of your nails, you can accurately diagnose the underlying chemical trauma and immediately intervene before the structural integrity of the entire nail bed is permanently compromised.
The following diagnostic list outlines the exact scientific correlations between visible physical symptoms and the underlying chemical damage caused by chronic alkaline exposure:
- Symptom: Deep horizontal depressions spanning the nail (Beau’s lines) = Cause: Acute matrix trauma from chemical burns temporarily shocking the system and halting keratin cell production.
- Symptom: Chronic, painful redness and throbbing swelling of the eponychium = Cause: Localized tissue liquefaction and severe contact dermatitis resulting from deep alkaline seepage.
- Symptom: The hard nail plate physically lifting away from the pink bed beneath (onycholysis) = Cause: Corrosive chemical pooling beneath the proximal nail fold that slowly dissolves the delicate hyponychium seal.
- Symptom: Rapid, aggressive regrowth of abnormally thick, jagged, and hard cuticle skin = Cause: The body’s defensive hyperkeratosis response desperately attempting to protect the compromised nail root from further chemical assaults.
- Symptom: Chalky, white, powdery patches appearing directly on the nail surface = Cause: Potassium hydroxide aggressively stripping the vital intracellular lipids from the top layers of the nail plate, causing severe dehydration and structural flaking often misdiagnosed as fungal infections.
Catching these aggressive symptoms early is the absolute difference between enduring a temporary cosmetic setback and fighting a lifelong battle with deformed, chronically painful fingernails.
Once the warning signs are evident, the immediate priority becomes halting the damage and carefully selecting products that respect your biological boundaries.
The Safe Path Forward: Recovery and Ingredient Intelligence
Rehabilitating a sensitive nail bed severely damaged by repeated, hidden chemical burns requires a complete and immediate overhaul of your home manicure routine. The fundamental focus must aggressively shift from rapid chemical removal to intensive, targeted hydration and physical, gentle micro-exfoliation. The living skin robustly surrounding your nails is absolutely not an enemy to be forcefully dissolved; rather, it is a vital, irreplaceable biological shield that demands careful preservation and structural respect.
The Top 3 Steps to Recover from Alkaline Burns
- Step 1: Complete Chemical Abstinence. Immediately inspect your cosmetic cabinets and discard any product listing Potassium hydroxide or sodium hydroxide in its top five active ingredients. You must allow the stratum corneum a full 28-day cellular turnover cycle to naturally rebuild its defensive acid mantle without any external alkaline interference.
- Step 2: Intensive Lipid Restoration. Apply a high-quality, cold-pressed jojoba oil or pure squalane serum directly to the eponychium twice daily. Jojoba oil is scientifically preferred because its microscopic molecular size closely mimics natural human sebum. Vigorously massage the oil in tight, circular motions for exactly sixty seconds per nail to massively stimulate localized blood flow and effectively deliver essential fatty acids directly to the recovering nail matrix.
- Step 3: Mechanical Micro-Exfoliation. Instead of chemically melting the skin, securely switch to safely softening the area by submerging your fingertips in warm water heated to roughly 98 degrees Fahrenheit for exactly five minutes. Following the soak, gently push back only the true, transparent dead cuticle tissue resting flat on the nail plate using a soft, silicone-tipped pusher, strictly avoiding dangerous, sharp metal implements that cause micro-tears.
| Product Component | What To Avoid (The Danger Zone) | What To Look For (The Safe Zone) |
|---|---|---|
| Active Softening Agent | Potassium hydroxide or Sodium hydroxide | Urea, Lactic acid, or gentle natural fruit enzymes |
| Overall pH Level | Highly alkaline formulations (pH 10.0 to 14.0) | Skin-compatible acidity mirroring natural barriers (pH 4.5 to 5.5) |
| Core Hydration Profile | Harsh, water-based formulas completely lacking any protective lipids | Rich, dense formulations containing raw Jojoba oil, pure Squalane, or vegetable Glycerin |
Transitioning away from corrosive chemicals will inevitably result in a brief, two-week period where the surrounding skin feels slightly thicker or rougher as it actively heals. However, dermatologists emphasize that this texture change is a definitive sign of positive cellular regeneration, not a grooming flaw. Embracing the profound science of nail anatomy genuinely empowers you to achieve a beautiful, clean aesthetic without ever sacrificing your long-term dermatological health.
By adopting these strict biological standards, you guarantee your hands remain both beautiful and structurally sound for every future manicure to come.